Biden Going Along With Trump's Gutting of Key Drug Price Controls
Summary
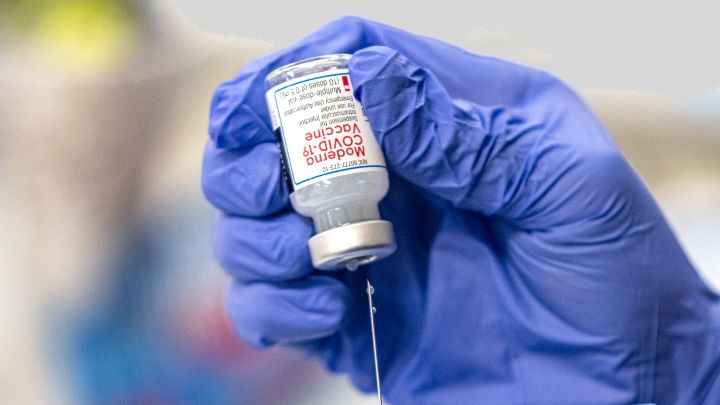
A last-minute Trump regulation would eliminate a key government authority that could be used against COVID vaccine price hikes.
Read This Post With a Free Trial
Unlock this post plus get our investigative reports with a two-week free trial subscription. Cancel anytime.
Already have an account? Sign in

